Home
We study early development using the zebrafish, as well as mouse, cell culture, and cavefish (Astyanax mexicanus) models.
A large component of our efforts are devoted to understanding mechanisms guiding the formation of blood and lymphatic vessels. The optical clarity and genetic and experimental accessibility of zebrafish make this a superb model organism for these studies. Our laboratory pioneered many of the key tools and resources used for vascular biology research in the zebrafish, including the widely used confocal microangiography method, a comprehensive atlas of the anatomy of the developing zebrafish vasculature, numerous vascular-specific transgenic fish lines, and methods for high resolution in vivo time-lapse imaging of zebrafish blood vessels. We have used these and other tools and resources to make a variety of seminal discoveries in the areas of vascular specification, differentiation, and patterning, including a novel pathway regulating arterial identity, a role for neuronal guidance factors in vascular patterning, a mechanism for vascular lumen formation in vivo, and identification and characterization of a lymphatic vascular system in the zebrafish. Cardiovascular-associated mortality is the leading cause of death in the western world, and many of the developmental processes we study are important in human congenital and acquired vascular diseases, as well as in cancer, heart disease, and ischemia.
We are also using cutting-edge molecular, cellular, genetic, transgenic, microscopic imaging, and next-gen sequencing approaches to study highly vascularized organs and tissues. This includes the meninges, the axillary lymphoid organ, and the gills. The meninges are complex, multilayered, highly vascularized tissues surrounding the brain that play a critical role in brain homeostasis and protection and are involved in a variety of brain pathologies. Our laboratory recently discovered that zebrafish have mammalian-like meninges readily observed and imaged in living animals through the thin, translucent adult zebrafish skull. We are using the advantages of the fish to study this critical but poorly understood set of tissues and to model meningeal cerebrovascular injury. We also recently discovered a previously uncharacterized lymph-node-like, externally located secondary immune organ, the “Axillary Lymphoid Organ” (ALO), that we are using to study how secondary immune organs develop. We have also carried out studies showing that the readily accessible zebrafish gills possess unusual gas-exchange endothelial cells that strongly resemble the specialized “aerocyte” endothelial cells of lung alveoli, and we are using the fish to study the development and function of these important cells.
Our laboratory has also recently developed an interest in studying the "genetics of epigenetics.” We have discovered novel epigenetic mechanisms regulating hematopoietic development and eye development, and we recently carried out the first large-scale forward-genetic screen in a vertebrate for tissue-specific epigenetic regulators using a novel “EpiTag” transgenic epigenetic reporter. This highly successful screen resulted in the identification of new mutants and genes that we are in the process of further characterizing and studying, including a liver-specific mutant that provides a new epigenetic model for metabolic-associated fatty liver disease (MAFLD).
Learn more about our recent research directions.
The Weinstein Laboratory is located in the intramural program of the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD), at the National Institutes of Health (NIH) in Bethesda, Maryland. The NIH Bethesda campus is located just outside Washington, D. C., and is the largest biomedical research institution in the world, with approximately 1200 principal investigators. Learn more about the NIH.
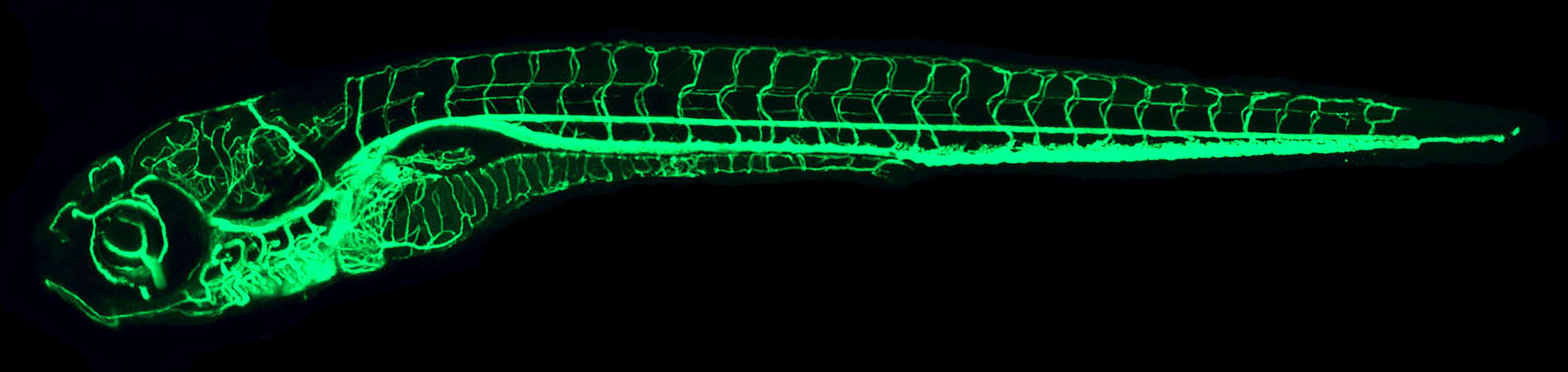
IMAGE: The confocal microangiography (intravascular injection of fluorescent dyes followed by confocal imaging) image above shows a lateral view of a 7 dpf whole zebrafish larva. The confocal microangiography technique was used by the Weinstein lab to compile a comprehensive atlas of vascular anatomy for the developing zebrafish.
Positions Available
Postdoctoral Positions - Angiogenesis and Lymphangiogenesis
Postdoctoral Positions - Studying Vascular-Associated Tissues and Organs
Postdoctoral Positions - Genetics of Epigenetics
To apply to positions send a cover letter, full resume, and other materials as noted to weinsteb@mail.nih.gov.
 BACK TO TOP
BACK TO TOP