Research
The Weinstein lab carries out studies on a diverse variety of innovative research topics, a few of which are noted below. The majority of the laboratory studies various aspects of blood and lymphatic vessel formation and biology, and the organization, function, and development of highly vascularized organs and tissues, but the lab has also recently pursued novel approaches to study the role of epigenetics during development. View a complete listing of our recent publications.
Studying the Vasculature and Vascular-Related Organs and Tissues
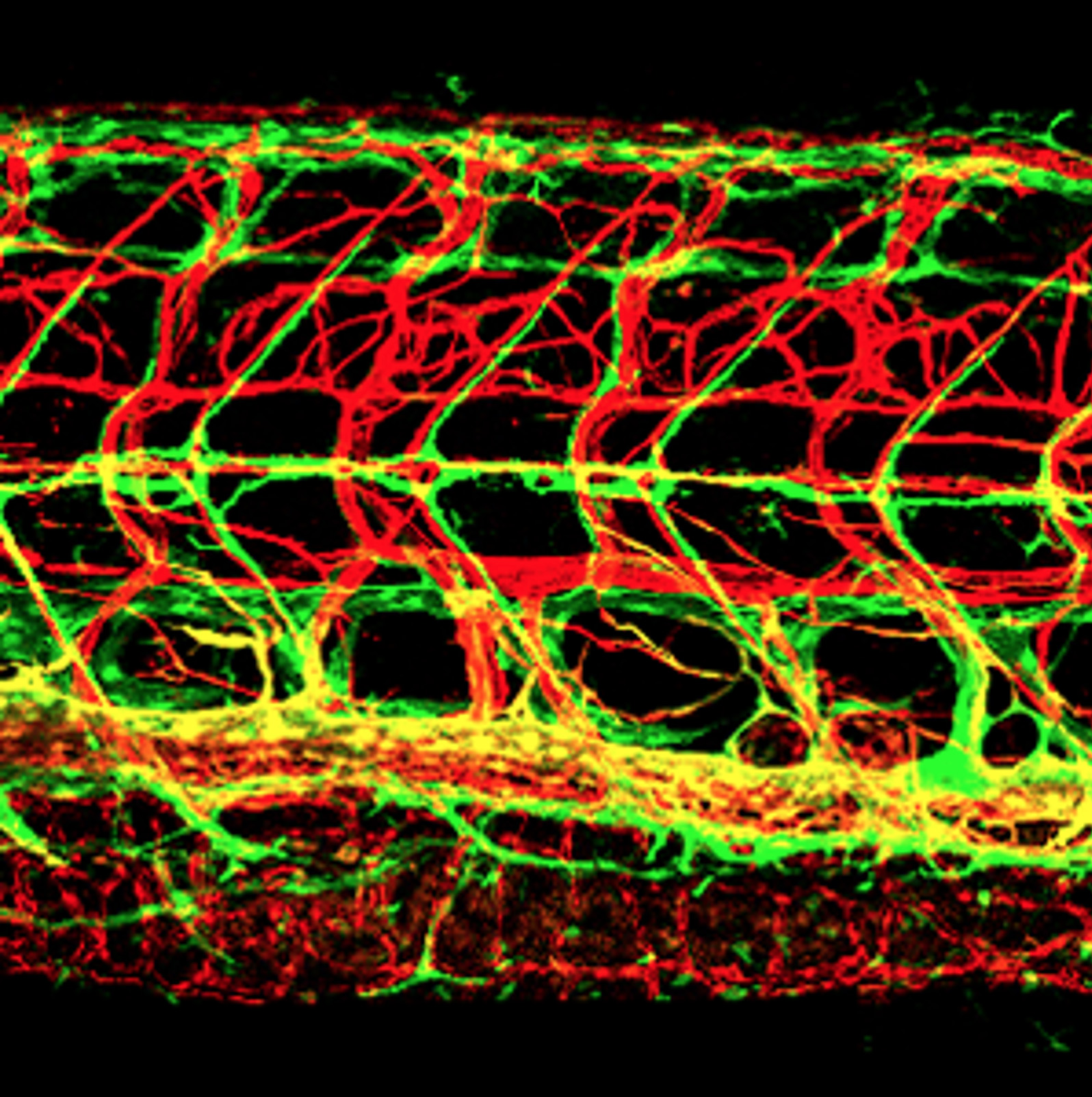 We seek to understand the cellular and molecular mechanisms responsible for the specification, patterning, and differentiation of blood and lymphatic vessels, how vascular networks arise during vertebrate embryogenesis, and how highly vascularized organs and tissues assemble and function. Blood vessels form a complex, highly ramified closed circulatory loop that innervates and supplies every tissue and organ with oxygen, nutrients, and cellular and humoral factors. Lymphatic vessels form a similarly complex but entirely separate highly branched, blind-ended network critical for fluid homeostasis, fat absorption, and immune function. Both types of vessels are involved in a variety of pathologies including vascular malformations, heart disease and ischemia, lung disease, and cancer. We are using the zebrafish as a genetically and experimentally accessible model for our studies, together with cell culture, mouse, and cavefish (Astyanax mexicanus) models.
We seek to understand the cellular and molecular mechanisms responsible for the specification, patterning, and differentiation of blood and lymphatic vessels, how vascular networks arise during vertebrate embryogenesis, and how highly vascularized organs and tissues assemble and function. Blood vessels form a complex, highly ramified closed circulatory loop that innervates and supplies every tissue and organ with oxygen, nutrients, and cellular and humoral factors. Lymphatic vessels form a similarly complex but entirely separate highly branched, blind-ended network critical for fluid homeostasis, fat absorption, and immune function. Both types of vessels are involved in a variety of pathologies including vascular malformations, heart disease and ischemia, lung disease, and cancer. We are using the zebrafish as a genetically and experimentally accessible model for our studies, together with cell culture, mouse, and cavefish (Astyanax mexicanus) models.
IMAGE: Lateral view confocal image of the trunk of a 12 dpf Tg(kdrl:cherry); Tg(mrc1a:egfp) double-transgenic zebrafish with red fluorescent blood vessels and green fluorescent lymphatics. See Jung et al. for more information. Linked movie shows 3-D rendering of a similar set of image data.
Vascular Specification and Patterning
Our laboratory has studied endothelial specification and arterial-venous or tissue-specific differentiation, the assembly of trunk and cranial blood and lymphatic vascular networks, and molecular cues that guide blood vessel and lymphatic vessel patterning, and extracellular and intracellular pathways that regulate blood vessel growth (including Rspo-Wnt and RECK-Wnt signaling, the Unfolded Protein Response (UPR) pathway, phosphoinositide recycling, and lipoprotein availability). We also recently characterized the function of the human lymphatic disease gene Shoc2.
The Vasculature in Wound Healing
We have developed new zebrafish models for studying how vessels contribute to healing, particularly in the context of skin abrasion wounds and meningeal cerebrovascular injury.
Two percent of the US population is plagued by poorly healing cutaneous wounds, most often in the elderly and diabetics. The reasons for poor healing remain unclear but vascular dysfunction is thought to be a major contributor. We developed a method to generate reproducible, discrete, readily survivable cutaneous abrasion wounds in adult zebrafish, currently being used to study the cellular and molecular drivers of cutaneous wound healing angiogenesis and how these go awry in aged or diabetic zebrafish.
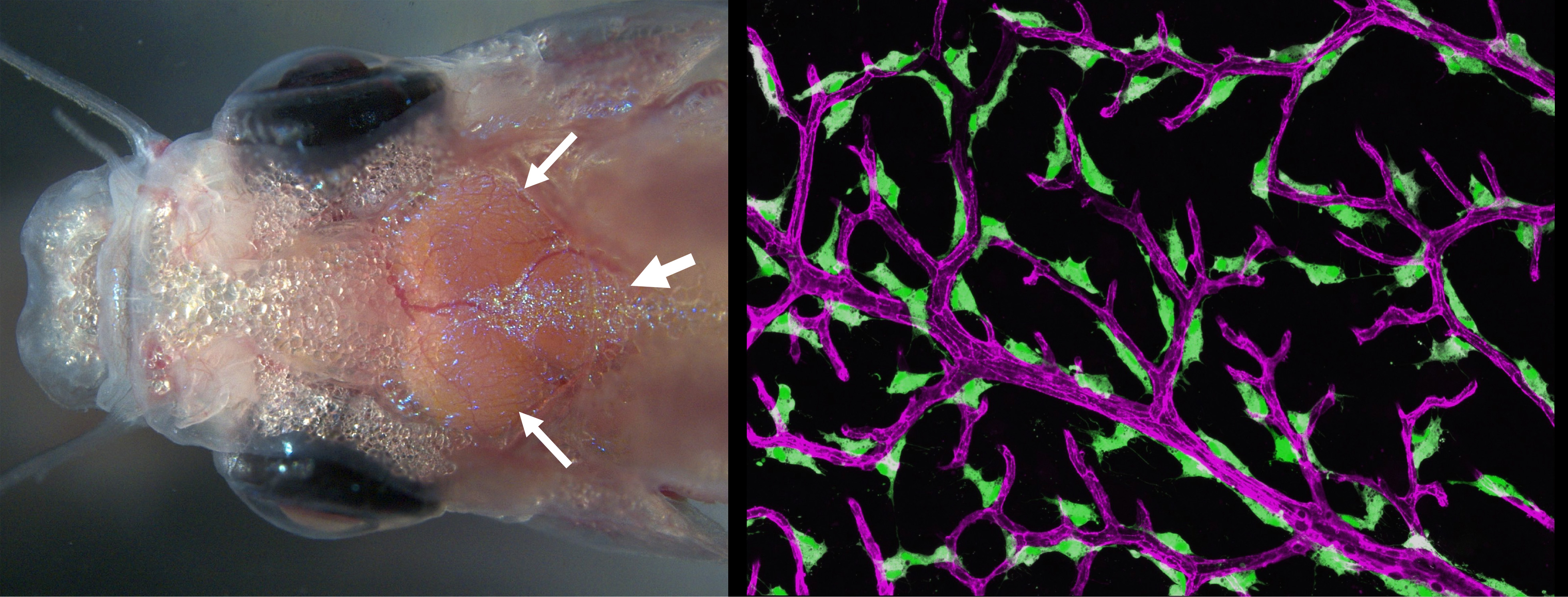
Traumatic head injuries affect nearly 1.4 million people in the United States each year. For many, prolonged bleeding and inflammation from damaged vessels in the meninges (thin, highly vascularized tissues that surround and protect the brain) result in long-lasting problems. Unlike mammals with thick, opaque skulls, adult zebrafish have a thin, translucent skull that allows for easy noninvasive imaging of the meninges in living animals. We recently showed that zebrafish have a mammalian-like meninges, making this a superb comparative model for longitudinal live imaging of the meninges. We have used these capabilities to help characterize a novel lymphatic-related perivascular scavenger cell residing in the leptomeningess. We recently established a new adult zebrafish meningeal cerebrovascular injury (mCVI) model using carefully calibrated sonication to damage meningeal blood vessels without breaching the skull or causing damage to the underlying brain. We are currently using this new model to observe vascular regrowth and immune responses to mCVI.
IMAGE: (LEFT) Dorsal view of the head of a living adult casper zebrafish with the surface of the brain clearly visible through the thin, translucent skull, including the two optic tecta (small arrows) and cerebellum (large arrow). (RIGHT) Confocal micrograph of "Fluorescent Granular Perithelial Cells" (FGPs, in green) adhering to the outside of meningeal blood vessels (in purple) on the brain of a Tg(mrc1a:egfp); Tg(kdrl:cherry) double-transgenic adult zebrafish. FGPs (aka muLECs) are unique endothelium-derived perivascular cells with unusual scavenging properties that are likely critical for brain homeostasis. The linked hi-res image shows an overview of FGPs (green) lining blood vessels (red) on the surface meninges of an adult zebrafish brain
Vascular-Associated Tissues and Organs
We are studying highly vascularized tissues and organs in the zebrafish, particularly those with strong human disease relevance. We recently described a previously uncharacterized external optically and experimentally accessible secondary immune organ in the zebrafish that we named the Axillary Lymphoid Organ or “ALO.” Lymph nodes and other secondary lymphoid organs play critical roles in immune surveillance and immune activation in mammals, but their deep internal locations make them challenging to image and study in living animals, and the origins and assembly of these important organs is still not well understood. The ALO is a small translucent organ located on the surface of zebrafish just above the pectoral fin. We showed that the ALO has a structure and cellular composition strongly reminiscent of lymph nodes and other secondary immune organs, making it a superb new model for live imaging of their assembly and function.
We have also been studying the zebrafish gills and their specialized vasculature as a comparative model for mammalian lungs. The high prevalence of lung disease makes it of compelling interest to uncover mechanisms to facilitate lung repair and regeneration, but the relative inaccessibility of mammalian lungs to observation and manipulation has made experimental study challenging. Unlike mammalian lungs, zebrafish gills are readily accessible to live imaging and experimental manipulation even in adults. We showed that gills possess many very similar cell types to lungs including unusual, highly specialized vascular endothelial cells resembling specialized “Aerocyte” endothelial cells found in mammalian lung alveoli. We are currently carrying out additional in-depth studies on these cells with an eye towards translational impact for human lung pathologies.
Genetic Analysis of Epigenetics
Our laboratory also studies the "genetics of epigenetics.” We discovered novel epigenetic mechanisms regulating hematopoietic development and eye development. We showed that the DNA methyltransferase DNMT3bb.1 regulates hematopoietic stem cell (HSC) maintenance by maintaining cmyb expression after an initial phase of runx1-dependent HSC specification. We also showed that excess DNA methylation-based epigenetic silencing promotes eye degeneration in blind Astyanax mexicanus cavefish morphs, and that DNA methylation-based gene repression can serve as an important molecular mechanism generating phenotypic diversity during development and evolution.
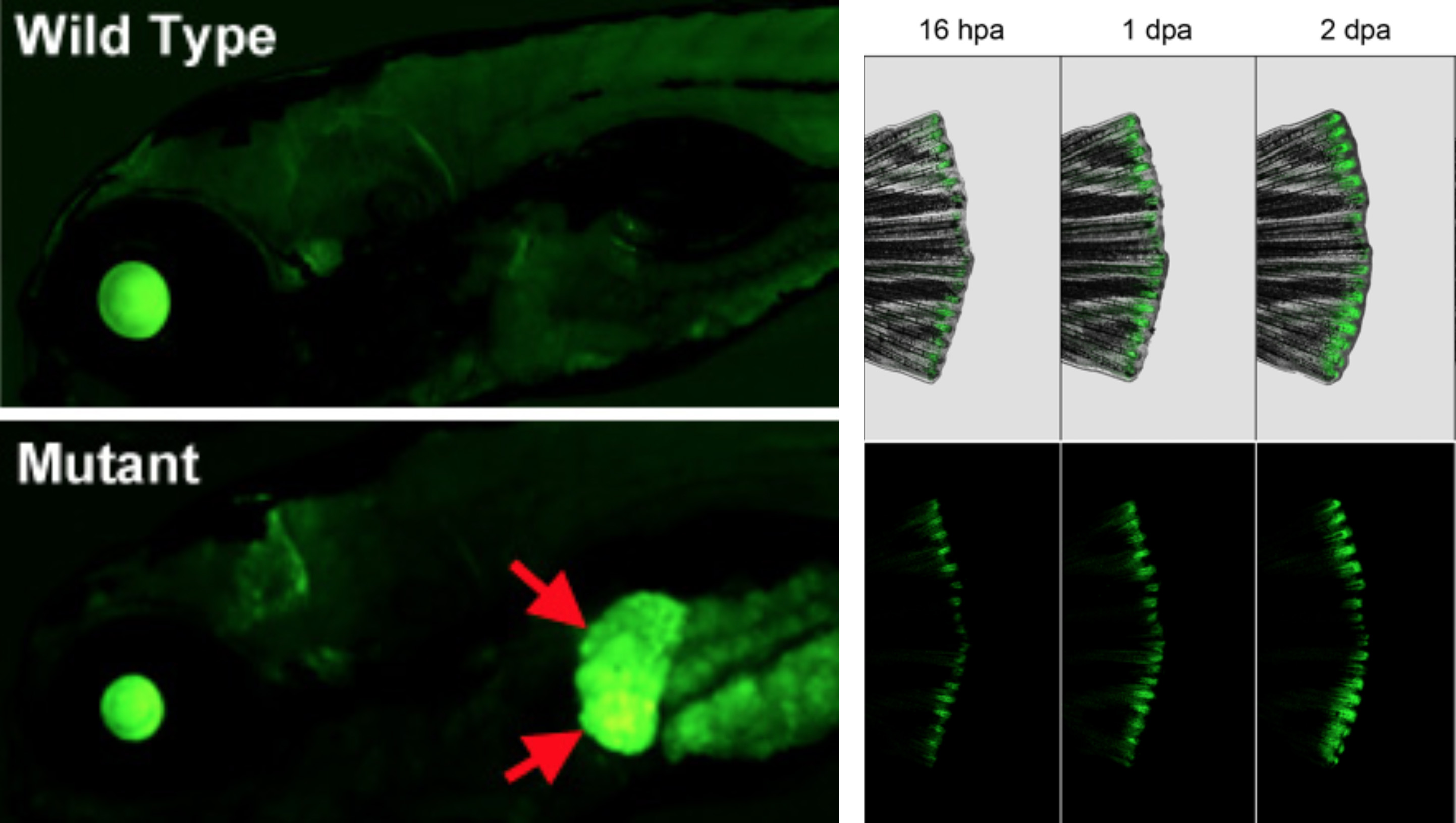
We recently developed a novel “EpiTag” zebrafish transgenic reporter line that permits real-time visualization of tissue-specific epigenetic silencing or activation in intact animals, making it possible for us to monitor dynamic changes in epigenetic regulation in living animals with cellular resolution. We used EpiTag fish to carry out the first large-scale forward-genetic screen in a vertebrate for tissue-specific epigenetic regulators. This highly successful ongoing screen resulted in identification of new mutants in epigenetic regulators, including a liver-specific epigenetic activation mutant that provides a new epigenetic model for metabolic-associated fatty liver disease (MAFLD), a disease affecting a quarter of the world’s population for which there is currently no approved drug therapy.
We are also using EpiTag transgenics to study the epigenetics of regeneration. The EpiTag reporter is activated in regenerating tissues and organs, including very early and striking activation in the regenerating fin. GFP activation serves as a highly specific marker for cells undergoing epigenetic reprogramming at otherwise inaccessible stages of regeneration, and we are using EpiTag expression to selectively enrich early-stage regenerating cells for multi-omic analysis of regeneration, leading to identification of novel epigenetic regulatory genes, molecular complexes, and pathways that we are currently further studying.
IMAGE: (LEFT) Lateral views of the head and anterior trunk of a wild type (top) and tissue-specific epigenetic silencing mutant (bottom) zebrafish. The mutant causes loss of epigenetic silencing specifically in the liver (red arrows), as visualized with the “EpiTag” transgenic reporter line developed in the Weinstein Lab. (RIGHT) Regenerating caudal fin of an EpiTag adult zebrafish at 16 hours, 1 day, and 2 days post-amputation. EpiTag expression is activated specifically in regenerating blastema tissue at the caudal margin of the fin.
Research Models and Methods
The zebrafish is the primary model organism for most of our studies, although we also use mice and especially endothelial cell culture in many of our projects, and use the Astyanax mexicanus surface and cavefish as a complementary aquatic model. We employ cutting-edge imaging and molecular technologies for our research work and we have developed a variety of important tools and methods to facilitate our studies. We also continue to use forward genetic screens to identify functionally critical genes of interest, using modern sequencing methods to facilitate rapid isolation of mutated genes.
Zebrafish
 Zebrafish are small tropical fish native to Southeast Asia that have a unique combination of genetic and experimental embryologic advantages making them ideal for studying early development, particularly the embryogenesis of the circulatory system. Learn more about zebrafish.
Zebrafish are small tropical fish native to Southeast Asia that have a unique combination of genetic and experimental embryologic advantages making them ideal for studying early development, particularly the embryogenesis of the circulatory system. Learn more about zebrafish.
Mexican Tetra (Astyanax mexicanus) Cavefish
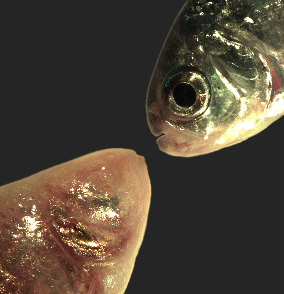 Astyanax mexicanus is a freshwater fish native to parts of southern Texas and eastern and central Mexico that exists in both surface-dwelling ("surface morphs") and very closely related cave-dwelling ("cave morphs") populations. Cave morphs have a series of uniquely evolved adaptations including loss of eyes and pigment, dramatically altered metabolism, altered vascular function, and altered sleep regulation and behavior. Results from our laboratory suggest that altered DNA methylation and resulting coordinated changes in expression of large sets of genes have helped to drive at least some of this rapid evolutionary change. Learn more about the Mexican Tetra Cavefish.
Astyanax mexicanus is a freshwater fish native to parts of southern Texas and eastern and central Mexico that exists in both surface-dwelling ("surface morphs") and very closely related cave-dwelling ("cave morphs") populations. Cave morphs have a series of uniquely evolved adaptations including loss of eyes and pigment, dramatically altered metabolism, altered vascular function, and altered sleep regulation and behavior. Results from our laboratory suggest that altered DNA methylation and resulting coordinated changes in expression of large sets of genes have helped to drive at least some of this rapid evolutionary change. Learn more about the Mexican Tetra Cavefish.
High-Resolution Imaging
 The externally developing, optically clear embryos and larvae of the fish make this a superb platform for high-resolution in vivo imaging. We employ a variety of sophisticated imaging modalities in our research work including "standard" confocal imaging, fast spinning disk confocal imaging, multi-photon imaging, super resolution (AiryScan) imaging, Selective Plane Illumination Microscopy (SPIM), and etc.. We have also developed numerous transgenic reporter lines and long-term time-lapse imaging methods to better take advantage of the favorable properties of fish and our imaging proficiency.
The externally developing, optically clear embryos and larvae of the fish make this a superb platform for high-resolution in vivo imaging. We employ a variety of sophisticated imaging modalities in our research work including "standard" confocal imaging, fast spinning disk confocal imaging, multi-photon imaging, super resolution (AiryScan) imaging, Selective Plane Illumination Microscopy (SPIM), and etc.. We have also developed numerous transgenic reporter lines and long-term time-lapse imaging methods to better take advantage of the favorable properties of fish and our imaging proficiency.
Genetic Screens
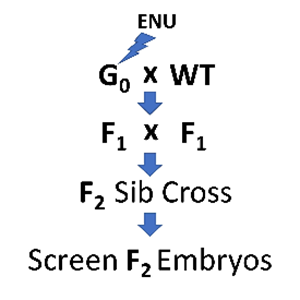 The ability to keep large numbers of fish and obtain large numbers of externally developing offspring make this a superb model for forward genetics. Although reverse-genetic "genome editing" has become popular amongst zebrafish researchers, and is used extensively in the Weinstein Lab, forward-genetic phenotype-based screens are still the most effective way to uncover genes essential for a particular process of interest. Molecular identification of the mutated genes is now fast and easy using next-gen sequencing methods to map mutants and identify causative lesions. We are expert in both genetic screens and in next-gen cloning of mutated genes.
The ability to keep large numbers of fish and obtain large numbers of externally developing offspring make this a superb model for forward genetics. Although reverse-genetic "genome editing" has become popular amongst zebrafish researchers, and is used extensively in the Weinstein Lab, forward-genetic phenotype-based screens are still the most effective way to uncover genes essential for a particular process of interest. Molecular identification of the mutated genes is now fast and easy using next-gen sequencing methods to map mutants and identify causative lesions. We are expert in both genetic screens and in next-gen cloning of mutated genes.
 BACK TO TOP
BACK TO TOP