Electrostatics of Membranes and Proteins
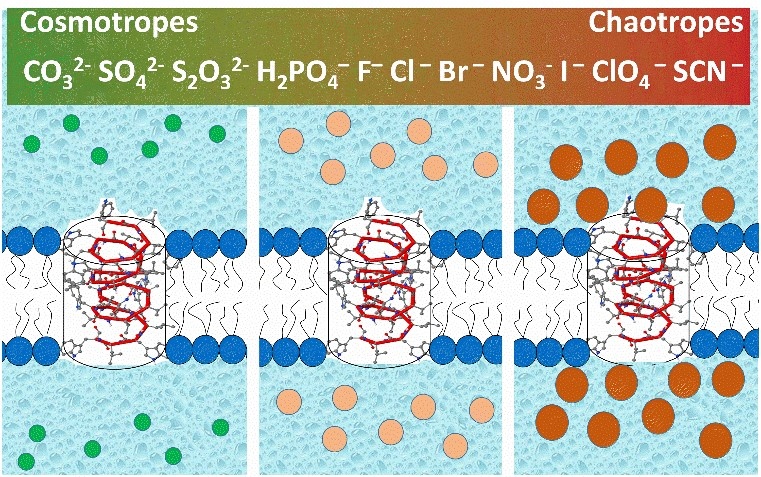
Long-range electrostatic forces are crucially involved in many interactions within and between biomolecules. Charged groups play the well-recognized roles in functioning of proteins, nucleic acids, phospholipids, and their supra-molecular assemblies. Phospholipid molecules, building blocks of cellular membranes, mostly contain zwitterionic or negatively charged headgroups exposed on the membrane/water interface. Membrane surface potential is an important determinant in regulation of membrane transport, cell-cell recognition, and membrane bound enzymes. We explore the functional consequences of electrostatics in channel-facilitated transport of metabolites by studying the changes in the channel properties upon (i) “salting out” the charges on the channel-forming molecules, (ii) varying the protonation state of channel residues by solution pH or changing the charge by point mutations, and (iii) modifying the surface charge of the membranes embedding these proteins by either changing membrane lipid composition and solution pH, adding multi-charge counterions, or using ions of different polarizability (Hofmeister effect).
Publications:
Rosencrans WM, Rajendran M, Bezrukov SM, and Rostovtseva TK (2021) VDAC regulation of mitochondrial calcium flux: From channel biophysics to disease. Cell Calcium. 94:102356. https://doi.org/10.1016/j.ceca.2021.102356 
Rosencrans WM, Aguilella VM, Rostovtseva TK, and Bezrukov SM (2021) alpha-Synuclein emerges as a potent regulator of VDAC-facilitated calcium transport. Cell Calcium. 95:102355. https://doi.org/10.1016/j.ceca.2021.102355 
Hoogerheide DP, Gurnev PA, Rostovtseva TK, and Bezrukov SM (2018) Real-Time Nanopore-Based Recognition of Protein Translocation Success. Biophys J. 1144:772-776. https://doi.org/10.1016/j.bpj.2017.12.019 
P.A. Gurnev, T.C. Roark, H.I. Petrache, A.J. Sodt, and S.M. Bezrukov. Cation-selective channel is regulated by anions according to their Hofmeister ranking. Angewandte Chemie, 2017, 56:3506–3509.
D.P. Hoogerheide, P.A. Gurnev, T.K. Rostovtseva, and S.M. Bezrukov. Mechanism of alpha-synuclein translocation through a VDAC nanopore revealed by energy landscape modeling of escape time distributions. Nanoscale, 2017, 9:183–192.
S.Y. Noskov, T.K. Rostovtseva, A.C. Chamberlin, O. Teijido, W. Jiang, and S.M. Bezrukov. Current state of theoretical and experimental studies of the voltage-dependent anion channel (VDAC). Biochimica et Biophysica Acta, 2016, 1858(7 Pt B):1778–1790.
S.M. Rappaport, O. Teijido, D.P. Hoogerheide, T.K. Rostovtseva, A.M. Berezhkovskii, and S.M. Bezrukov. Conductance hysteresis in the voltage-dependent anion channel. European Biophysics Journal, 2015, 44:465–472.
O. Teijido, S.M. Rappaport, A. Chamberlin, S.Y. Noskov, V.M. Aguilella, T.K. Rostovtseva, S.M. Bezrukov. Acidification asymmetrically affects voltage-dependent anion channel implicating the involvement of salt bridges. Journal of Biological Chemistry, 2014, 289:23670–23682.
P.A. Gurnev, T.L. Yap, C.M. Pfefferkorn, T.K. Rostovtseva, A.M. Berezhkovskii, J.C. Lee, V.A. Parsegian, and S.M. Bezrukov. Alpha-synuclein lipid-dependent membrane binding and translocation through the alpha-hemolysin channel. Biophysical Journal, 2014, 106:556–565.
S.Y. Noskov, T.K. Rostovtseva, and S.M. Bezrukov. ATP transport through VDAC and VDAC/tubulin complex probed by equilibrium and non-equilibrium MD simulations. Biochemistry, 2013, 52:9246−9256.
P.A. Gurnev, S.-T. Yang, K.C. Melikov, L.V. Chernomordik, and S.M. Bezrukov. Cationic cell-penetrating peptide binds to planar lipid bilayers containing negatively charged lipids but does not induce conductive pores. Biophysical Journal, 2013, 104:1933–1939.
P.A. Gurnev and S.M. Bezrukov. Inversion of membrane surface charge by trivalent cations probed with a cation-selective channel. Langmuir, 2012, 28:15824-15830.
E.M. Nestorovich, V.A. Karginov, A.M. Berezhkovskii, V.A. Parsegian, and S.M. Bezrukov. Kinetics and thermodynamics of binding reactions as exemplified by anthrax channel blockage with a cationic cyclodextrin derivative. Proc. Natl. Acad. Sci. USA, 2012, 109:18453–18458.
S.M. Bezrukov, X. Liu, V.A. Karginov, A.N. Wein, S.H. Leppla, M.R. Popoff, H. Barth, and E.M. Nestorovich. Interactions of high-affinity cationic blockers with the translocation pores of B. anthracis, C. botulinum, and C. perfringens binary toxins. Biophysical Journal, 2012, 103:1208–1217.
T.K. Rostovtseva, P.A. Gurnev, M.-Y. Chen, S.M. Bezrukov. Membrane lipid composition regulates tubulin interaction with mitochondrial voltage-dependent anion channel. Journal of Biological Chemistry, 2012, 287:29589–29598.
P.A. Gurnev, R. Ortenberg, T. Dörr, K. Lewis, and S.M. Bezrukov. Persister-promoting bacterial toxin TisB produces anion-selective pores in planar lipid bilayers. FEBS Letters, 2012, 586:2529–2534.
P.A. Gurnev, M. Queralt-Martin, T.K. Rostovtseva, V.M. Aguilella, and S.M. Bezrukov. Probing tubulin-blocked state of VDAC by varying membrane surface charge. Biophysical Journal, 2012, 102:2070–2076.
T.K. Rostovtseva and S.M. Bezrukov. VDAC interaction with tubulin and its physiological implications. Biochimica et Biophysica Acta, 2012, 1818:1526–1535.
E.M. Nestorovich, V.A. Karginov, and S.M. Bezrukov. Polymer partitioning and ion selectivity suggest asymmetrical shape for the membrane pore formed by Epsilon toxin. Biophysical Journal, 2010, 99:782–789.
E.M. Nestorovich, V.A. Karginov, A.M. Berezhkovskii, and S.M. Bezrukov. Blockage of anthrax PA63 pore by a multi-charged high-affinity toxin inhibitor. Biophysical Journal, 2010, 99:134–143.
A. Alcaraz, E.M. Nestorovich, M.L. López, E. García-Gimenez, S.M. Bezrukov, and V.M. Aguilella. Diffusion, exclusion, and specific binding in a large channel: A study of OmpF selectivity inversion. Biophysical Journal, 2009, 96:56–66.
C. Danelon, E.M. Nestorovich, M. Winterhalter, M. Ceccarelli, and S.M. Bezrukov. Interaction of zwitterionic penicillins with the OmpF channel facilitates their translocation. Biophysical Journal, 2006, 90:1617–1627.
A. Alcaraz, E.M. Nestorovich, M. Aguilella-Arzo, V.M. Aguilella, and S.M. Bezrukov. Salting out the ionic selectivity of a wide channel: The asymmetry of OmpF. Biophysical Journal, 2004, 87:943–957.
E.M. Nestorovich, T.K. Rostovtseva, and S.M. Bezrukov. Residue ionization and ion transport through OmpF channels. Biophysical Journal, 2003, 85:3718–3729.
E.M. Nestorovich, C. Danelon, M. Winterhalter, and S.M. Bezrukov. Designed to penetrate: Time-resolved interaction of single antibiotic molecules with bacterial pores. Proc. Natl. Acad. Sci. USA, 2002, 99:9789–9794.
V.V. Malev, L.V. Schagina, P.A. Gurnev, J.Y. Takemoto, E.M. Nestorovich, and S.M. Bezrukov. Syringomycin E channel: A lipidic pore stabilized by lipopeptide? Biophysical Journal, 2002, 82:1985–1994.
S.M. Bezrukov and J.J. Kasianowicz. Neutral polymers in the nanopores of alamethicin and alpha-hemolysin. Biol. Membrany, 2001, 18:453–457. (PDF - 93.4KB)
V.M. Aguilella and S.M. Bezrukov. Alamethicin channel conductance modified by lipid charge. European Biophysics Journal, 2001, 30:233–241.
V.V. Malev, Y.A. Kaulin, P.A. Gurnev, S.M. Bezrukov, J.Y. Takemoto, and L.V. Schagina. Effects of charge distribution on conductance of single channels formed by syringomycin E in lipid bilayers. Biol. Membrany, 2001, 18:145-153 (in Russian).
S.M. Bezrukov, R.P. Rand, I. Vodyanoy, and V.A. Parsegian. Lipid packing stress and polypeptide aggregation: alamethicin channels probed by proton titration of lipid charge. Faraday Discussions, 1998, 111:173-183.
T.K. Rostovtseva, V.M. Aguilella, I. Vodyanoy, S.M. Bezrukov, and V.A. Parsegian. Membrane surface charge titration probed by gramicidin A channel conductance. Biophysical Journal, 1998, 75:1783-1792.
S.M. Bezrukov and J.J. Kasianowicz. The charge state of an ion channel controls neutral polymer entry into its pore. 
J.J. Kasianowicz and S.M. Bezrukov. Protonation dynamics of the alpha-toxin channel from spectral analysis of pH dependent current fluctuations. Biophysical Journal, 1995, 69:94-105.
S.M. Bezrukov and J.J. Kasianowicz. Current noise reveals protonation kinetics and number of ionizable sites in an open protein ion channel. Physical Review Letters, 1993, 70:2352-2355.
 BACK TO TOP
BACK TO TOP