NICHD Ovarian Nomenclature Workshop 2021
Investigation into the ovary spurred by OTC, has demonstrated that our traditional description of the ovarian anatomy is inadequate as research in the field progresses. One obstacle to the advancement of knowledge in female fertility and reproduction is that there is a paucity of distinction between the different cellular and anatomical compartments of the ovary. In May-June 2021, Dr. Gomez-Lobo convened scientists from around the U.S. for several Division of Intramural Research (DIR) supported Ovarian Tissue Nomenclature Workshop meetings. Experts in reproductive endocrinology, gynecologic oncology, radiology, pathology, bioinformatics, and ovarian tissue research, met and created workgroups to further delineate ovarian follicle description, tissue ontology, and the 3-D description of the ovary. Resulting recommendations will establish the new national and international classification standards for ovary research which will allow for clearer evaluation of the function of the ovary at a gross and sub-anatomic level.
Workshop recommendations:
Workshop Sub-Groups
Gross Anatomy Workgroup Recommendations
The Ovarian Gross Anatomy Working Group recommended use of a three-dimensional (3D) regional model of the ovary based on the organ’s macroscopically visible structures and major points of fixation.
Sub-anatomy Workgroup Recommendations
The Subanatomy Working Group reviewed the current ontology of the ovary and proposed updates to the Uberon classification by referencing histologic features with a revised list of subanatomic structures, including cell types and extracellular features.
![]()
Follicle Workgroup Recommendations
The Follicle Classification Working Group recommended the expansion of the current classification system to include six stages of preantral follicles, five stages of antral follicles, three categories of abnormal preantral follicles, abnormal antral follicles, multi-oocytic follicles, and follicle variations of unknown significance (FVUS).
Table 1. Classification System of Human Ovarian Follicle Morphology
| Follicle Classes | Morphologic Characteristics | ||
|---|---|---|---|
| Preantral Follicle | Preantral Follicles with Normal Morphology | Primordial ovarian follicle | Oocyte surrounded by a continuous or an incomplete single layer of squamous granulosa cells. Follicle diameter ~ 40-44 μm; oocyte diameter ~ 29-35 μm*1-5 |
| Transitional primordial ovarian follicle | Oocyte surrounded by a continuous or incomplete single layer consisting of both squamous and cuboidal granulosa cells.6, 7 Follicle diameter ~ 44 μm; oocyte diameter ~ 36 μm*1 | ||
| Primary ovarian follicle | Oocyte surrounded by a complete single layer of cuboidal granulosa cells. Follicle diameter ~ 41-100 μm; oocyte diameter ~ 29-40 μm*1-4,8 | ||
| Transitional primary ovarian follicle | Oocyte surrounded by reorganizing cuboidal granulosa cells that exist in a partial single and double layer; zona pellucida and theca layer may be visible.6,7,9 | ||
| Secondary ovarian follicle | Oocyte surrounded by 2-3 layers of cuboidal granulosa cells surrounded by the theca interna layer. Zona pellucida present. Follicle diameter ~ 56-200 μm; oocyte diameter ~ 35-55 μm*1-3,8 | ||
| Multilayer ovarian follicle | Oocyte surrounded by >3 layers of cuboidal granulosa cells with outer layer of theca cells. Zona pellucida present. Follicle diameter ~250-300 μm; oocyte diameter 100-120 μm.4,10 | ||
| Preantral Follicle with Abnormal Morphological Features | Preantral Follicles with abnormal morphological features of oocyte [AMF-o] | Preantral follicles with vacuoles in oocyte and/or containing degenerating oocytes. | |
| Preantral Follicles with abnormal morphological features of granulosa cells [AMF-g] | Preantral follicles with vacuoles in granulosa cells and/or containing degenerating granulosa cells. | ||
| Preantral Follicles with abnormal morphological features in both oocytes and granulosa cells [AMF-og] | Preantral follicles with vacuoles in both oocytes and granulosa cells and/or containing both degenerating oocytes and granulosa cells. | ||
| Antral Follicle | Antral Follicles with Normal Morphology | Cumulus-enclosed oocyte in a fluid-filled space (antrum) surrounded by multiple layers of mural granulosa cells separated by a basement membrane from the adjacent layer of theca interna cells and the outermost layer of theca externa cells.10 Measured either histologically or on ultrasound. Human follicle has the ability to resume meiosis, with a follicle diameter 2-5mm and oocyte diameter ~ 100-120 μm.11,12 Minimal follicle diameter for developmental competence ~5-7mm.13 | |
| Early antral | Antral follicle with antrum diameter < 2 mm on histology.5 Follicle diameter ~250-400 μm; oocyte diameter 100-120 μm2-4,11 | ||
| Pre-selection | Antral follicle with antrum diameter 2-5 mm on imaging (possibly histology**)5 | ||
| Selection | Antral follicle with antrum diameter 6-9 mm on imaging (possibly histology**)14 | ||
| Dominance | Antral follicle with antrum diameter 10-15 mm on imaging (possibly histology**)5-17 | ||
| Pre-ovulatory | Antral follicle with antrum diameter 16-28 mm on imaging (possibly histology**). Human oocyte diameter ~ 100-140 μm.2,11,18 | ||
| Atretic Antral Follicle with Normal Morphology | Antral follicle of any stage wherein granulosa cells proximal to the antrum detach from the mural layers that appear as single or multiple cells within the antrum; these granulosa cells typically have pyknotic nuclei; follicle may contain an oocyte with abnormal morphology. | ||
| Antral Follicle with Abnormal Morphological Features | Antral follicles containing a subset of atretic-appearing granulosa cells enclosing an oocyte with normal or abnormal morphology. | ||
| Corpus Luteum (CL) and derivatives | CL of the Menstrual Cycle | Grows to ~1.5cm diameter. It contains 1) luteal tissue granulosa-lutein layer plus the theca-lutein area; 2) inner boundary of connective tissue; 3) central cavity that was filled with follicular fluid or blood.19 There is a combination of small and large luteal cells, each with a round, centrally located nucleus and prominent nucleolus.20 The luteal cells are surrounded by a connective tissue capsule and is adjacent to a highly vascularized capillary network. Fibroblast cells, collagen fibers, endothelial cells and macrophages are present. Variations of cell composition, vascularity, and quantity of collagen occur based on timing within the luteal phase. As the CL undergoes luteolysis and loses its ability to produce progesterone, there are degenerative changes, cytoplasmic vacuolization, macrophage marker expression, leading to apoptosis.19 Limited data in humans, different stages noted in the Buffalo.20 | |
| CL of Pregnancy | Same histologic appearance as CL of the menstrual cycle. (Above). Limited data.20 | ||
| Corpora Albicans | Small white scars typically present in the medulla that consist of collagen Type I from fibroblasts. Generally separated from the cortex by a thin band of medullary stromal cells. | ||
| Hemorrhagic Anovulatory Follicle | An inner lining of fibrous tissue (2–8 layers thick) surrounded by luteinized cells (up to 50 layers thick) Arterioles and venules, extended into the center of the follicle with a central cavity filled with red blood cells. Seen in humans21 and mammals22. | ||
| Luteinized Unruptured Follicle | Oocytes trapped within CLs and expanded cumulus oocyte complexes within unruptured antral follicles. Found in reproductive humans3-25 and mammals26-28 | ||
| Multi-oocytic follicles | ≥2 oocytes surrounded by squamous or cuboidal granulosa cells; represent germ cell cysts that have failed to breakdown. | ||
| Follicle Variations of Unknown Significance (FVUS) | Follicles that do not fit any above definition; i.e. large primordial follicles as observed in prepubertal ovaries. | ||
| Tangential | Cluster of granulosa cells without an oocyte indicating a follicle edge, but unable to distinguish stage of follicle. | ||
*Size of follicle and oocyte may vary depending on fresh or frozen tissue as well as processing techniques that potentially can cause shrinkage.
**Antral follicles can be seen histologically. The accuracy of the size may vary depending on whether the tissue is fresh or frozen. Follicle may rupture or become distorted during tissue biopsy, fixation, and sectioning, therefore altering the diameter of the antrum. Histology measurements may not match ultrasound measurements. Oocytes may not be seen since some are aspirated or extruded into the media during processing for ovarian tissue cryopreservation.
Table 2. Abnormal morphological features sometimes observed in follicles29,30
| Vacuoles or spaces inside the oocyte, although the granulosa cells look healthy |
| Large spaces between the granulosa cells, but the oocyte appears normal |
| Dissociated granulosa cells and abnormal oocyte |
| Large vacuoles in the oocyte |
| Misshapen oocyte with normal granulosa cell layers |
| Degenerate oocyte and pyknotic granulosa cells |
Figure 1: Follicle Nomenclature H&E Stained Examples
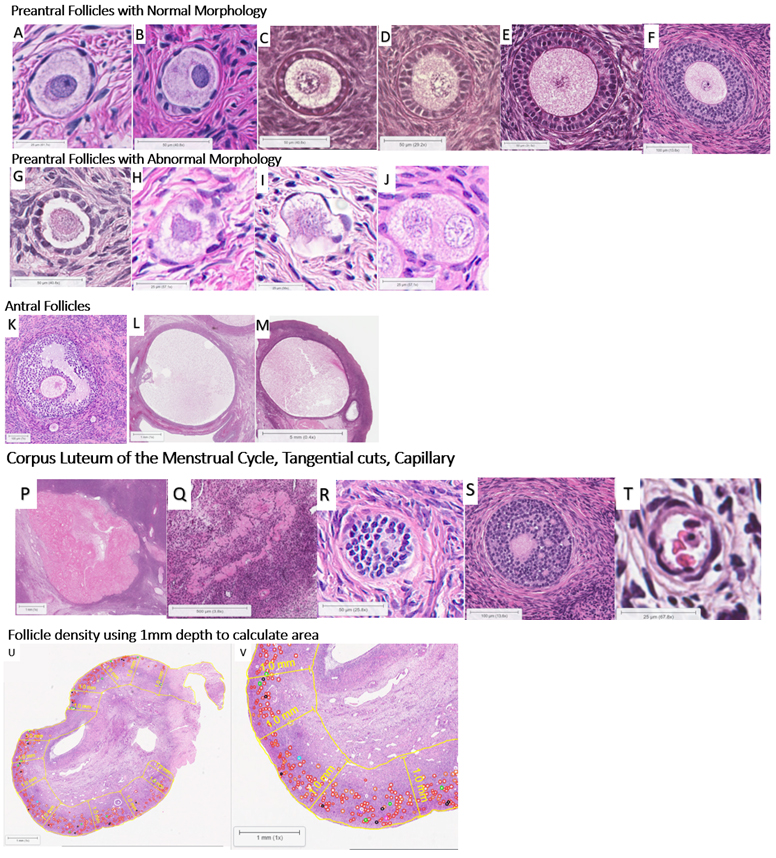
Figure 1: A. Primordial: Oocyte surrounded by a continuous or an incomplete single layer of squamous granulosa cells. B. Transitional Primordial: Oocyte surrounded by a continuous or incomplete single layer consisting of both squamous and cuboidal granulosa cells. C. Primary: Oocyte surrounded by a complete single layer of cuboidal granulosa cells. D. Transitional Primary: Oocyte surrounded by reorganizing cuboidal granulosa cells in a partial single and double layer; zona pellucida and theca layer may be visible. E. Secondary: Oocyte surrounded by 2-3 layers of cuboidal granulosa cells with outer layer of theca cells. F. Multilayer: Oocyte surrounded by >3 layers of cuboidal granulosa cells with outer layer of theca cells.G. Abnormal Morphological features of the oocyte (AMF-o): Vacuoles in oocyte and/or containing degenerating oocytes. H. Abnormal Morphological features of granulosa cells (AMF-g): Vacuoles in granulosa cells and/or containing degenerating granulosa cells. I. Abnormal morphological features in both oocytes and gcs (AMF-og): Vacuoles in both oocytes and granulosa cells and/or containing both degenerating oocytes and granulosa cells. J. Multi-oocytic: ≥2 oocytes surrounded by squamous or cuboidal granulosa cells; represent germ cell cysts that have failed to breakdown. K: Early Antral Follicle (<2mm diameter). L: Pre-selection Antral Follicle (2-5mm diameter). M: Selection Antral Follicle (6-9 mm diameter). N: Atretic Antral with Normal Morphology. O: Atretic Antral with Abnormal Morphology. P: Corpus Luteum. Q: Corpus Albicans. R: Tangential cut. S: Tangential antral follicle cut. T: Capillary. U: Cross section of ovary. Example of calculating follicle density by using 1mm depth of the cortex to calculate area. V: Zoomed in view of cross section of ovary.
Figure 2: Follicle morphology comparison with different fixatives.
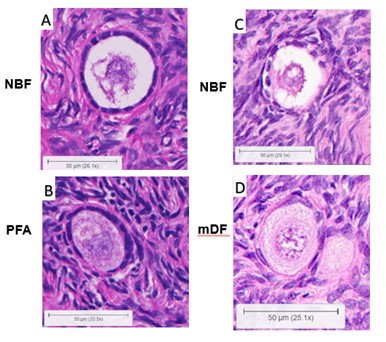
Figure 2A and 2B are from the same human ovary (Patient 1), comparing 10% Neutral Buffered Formalin (NBF) vs. 4% paraformaldehyde (PFA). Figure 2C and 2D are from the same human ovary (Patient 2), comparing NBF vs. modified Davidson’s Fixative (mDF).
Box 1: Suggested Follicle Density Guidelines:
| SECTIONS AND AREA |
|---|
| Section using 5μm |
| Count every 5th section |
| Area for Follicle density |
| Use area that is 1mm in depth from the edge of the cortex |
| Calculate follicles/mm2 |
| Average follicles/mm<2 based on the number of sections |
| FOLLICLE COUNTING QUALIFICATIONS |
|---|
| Primordial/ Transitional Primordial: Count all* |
| Primary/ Transitional Primary: Count all* |
| Secondary/Multilayer: Count only ones with oocyte nucleus present |
| All Antral Stages: Count only ones with oocyte nucleus present |
| Count abnormal follicles separately - AMF-o, AMF-g, AMF-og, multioocytic, and FVUS |
*If there is only one section to analyze, follicle counting guidelines include counting all Primordial, Transitional Primordial, Primary, and Transitional Primary regardless of whether the oocyte nucleus is present or absent. If there is more than 1 section available, only count if the oocyte nucleus is present to avoid double counting of follicles because the average diameter of an oocyte in a primordial follicle is ~ 29-35 μm.*1-5
Workshop Participants
| Name | Affiliation |
|---|---|
| Christina Annunziata, MD, PhD | National Cancer Institute |
| G Thomas Brown, MD, PhD | Artificial Intelligence Resource; National Cancer Institute |
| Alan H DeCherney, MD | Reproductive Endocrinology and Infertility; Eunice Kennedy Shriver National Institute of Child Health and Human Development |
| Tazim Dowlut-McElroy, MD | Pediatric and Adolescent Gynecology Program; Eunice Kennedy Shriver National Institute of Child Health and Human Development |
| Francesca E Duncan, PhD | Department of Obstetrics and Gynecology; Feinberg School of Medicine, Northwestern University |
| Veronica Gomez-Lobo, MD | Pediatric and Adolescent Gynecology Program; Eunice Kennedy Shriver National Institute of Child Health and Human Development |
| Clarisa R Gracia, MD | Division of Reproductive Endocrinology and Infertility/Fertility Preservation; University of Pennsylvania Perelman School of Medicine |
| Stefan Gysler, MD | Division of Gynecologic Oncology; University of Pennsylvania Perelman School of Medicine |
| Lisa M Halvorson, MD | USMA-Bayer Pharmaceuticals (Current Affiliation) Affiliation at time of workshop: Gynecologic Health and Disease Branch; Eunice Kennedy Shriver National Institute of Child Health and Human Development |
| Tracy Harrison, MD | Reproductive Sciences; University of California San Diego |
| Junhyong Kim, PhD | University of Pennsylvania Perelman School of Medicine |
| Monica M Laronda, PhD | Stanley Manne Children's Research Institute, Ann & Robert H Lurie Children's Hospital of Chicago, Feinberg School of Medicine, Northwestern University |
| Richard D LeDuc, PhD | Children's Hospital Research Institute of Manitoba |
| Hong Lou, MD | Pediatric and Adolescent Gynecology Program; Eunice Kennedy Shriver National Institute of Child Health and Human Development |
| Marla Lujan, PhD | Division of Nutritional Sciences; Cornell University |
| Jacqueline Y Maher, MD | Pediatric and Adolescent Gynecology Program; Eunice Kennedy Shriver National Institute of Child Health and Human Development |
| Kathleen O’Neill, MD, MSTR | Division of Reproductive Endocrinology and Infertility; University of Pennsylvania Perelman School of Medicine |
| Kutluk H Oktay, MD, PhD | Department of Obstetrics, Gynecology, and Reproductive Sciences; Yale University School of Medicine |
| Stephanie A Pangas, PhD | Department of Pathology and Immunology; Baylor College of Medicine |
| Alison Pouch, PhD | Departments of Radiology & Bioengineering: University of Pennsylvania Perelman School of Medicine |
| Rebecca Rakow-Penner, MD, PhD | Department of Radiology; University of California San Diego |
| Julie Rios, MD | Reproductive Endocrinology and Infertility; University of Pittsburgh (Present affiliation) |
| Erin Rowell, MD | Associate Professor of Surgery; Northwestern University Feinberg School of Medicine |
| Lauren E Schwartz, MD | Department of Pathology; University of Pennsylvania Perelman School of Medicine |
| James Segars, MD | Reproductive Science and Women’s Health Research; Johns Hopkins University |
| Ariella Shikanov, PhD | Department of Biomedical Engineering, Department of Obstetrics and Gynecology; University of Michigan |
| Susan Taymans, PhD | Eunice Kennedy Shriver National Institute of Child Health and Human Development, National Institutes of Health |
| Candace Tingen, PhD | Gynecologic Health and Disease Branch; Eunice Kennedy Shriver National Institute of Child Health and Human Development, National Institutes of Health |
| Elizabeth L Tsui, BS | Stanley Manne Children's Research Institute, Ann & Robert H Lurie Children's Hospital of Chicago, Feinberg School of Medicine, Northwestern University |
| Mary B Zelinski, PhD | Oregon National Primate Research Center; Oregon Health & Science University |
Publications
- Yano Maher JC, Zelinski MB, Oktay KH, Duncan FE, Segars JH, Lujan ME, Lou H, Yun B, Hanfling SN, Schwartz LE, Laronda MM, Halvorson LM, O'Neill KE, Gomez-Lobo V. Classification of Human Ovarian Follicle Morphology: Recommendations of the NICHD-Sponsored Ovarian Nomenclature Workshop. Fertil Steril. 2024 Nov 14:S0015-0282(24)02394-X. doi: 10.1016/j.fertnstert.2024.11.016. Epub ahead of print. PMID: 39549739.
- Tsui EL, O'Neill KE, LeDuc RD, Shikanov A, Gomez-Lobo V, Laronda MM. Creating a common language for the subanatomy of the ovary. Biol Reprod. 2022 Oct 29:ioac199. doi: 10.1093/biolre/ioac199. Epub ahead of print. PMID: 36308436.
- O'Neill KE, Maher JY, Laronda MM, Duncan FE, Leduc RD, Lujan ME, Oktay KH, Pouch AM, Segars JH, Tsui EL, Zelinski MB, Halvorson LM, Gomez-Lobo V. Anatomic Nomenclature and 3D Regional Model of the Human Ovary: Call for a New Paradigm. Am J Obstet Gynecol. 2022 Sep 30:S0002-9378(22)00795-5. doi: 10.1016/j.ajog.2022.09.040. Epub ahead of print. PMID: 36191605.
References
- Westergaard CG, Byskov AG, Andersen CY. Morphometric characteristics of the primordial to primary follicle transition in the human ovary in relation to age. Hum Reprod. Aug 2007;22(8):2225-31. doi:10.1093/humrep/dem135
- Jiang Y, He Y, Pan X, Wang P, Yuan X, Ma B. Advances in Oocyte Maturation In Vivo and In Vitro in Mammals. Int J Mol Sci. May 21 2023;24(10)doi:10.3390/ijms24109059
- Erickson GF. The ovary: Basic principles and concepts. Endocrinology and Metabolism. McGraw-Hill; 1995.
- Griffin J, Emery BR, Huang I, Peterson CM, Carrell DT. Comparative analysis of follicle morphology and oocyte diameter in four mammalian species (mouse, hamster, pig, and human). J Exp Clin Assist Reprod. Mar 1 2006;3:2. doi:10.1186/1743-1050-3-2
- Gougeon A. Human ovarian follicular development: from activation of resting follicles to preovulatory maturation. Ann Endocrinol (Paris). May 2010;71(3):132-43. doi:10.1016/j.ando.2010.02.021
- Oktay K, Schenken RS, Nelson JF. Proliferating cell nuclear antigen marks the initiation of follicular growth in the rat. Biology of reproduction. Aug 1995;53(2):295-301. doi:10.1095/biolreprod53.2.295
- Muskhelishvili L, Wingard SK, Latendresse JR. Proliferating cell nuclear antigen--a marker for ovarian follicle counts. Toxicol Pathol. 2005;33(3):365-8. doi:10.1080/01926230590930164
- Quan N, Mara JN, Grover AR, Pavone ME, Duncan FE. Spatial Analysis of Growing Follicles in the Human Ovary to Inform Tissue Engineering Strategies. Tissue Eng Part A. Jul 2020;26(13-14):733-746. doi:10.1089/ten.tea.2020.0051
- Oktay KH, Oktay MH. Immunohistochemical analysis of tyrosine phosphorylation and AP-1 transcription factors c-Jun, Jun D, and Fos family during early ovarian follicle development in the mouse. Appl Immunohistochem Mol Morphol. Dec 2004;12(4):364-9. doi:10.1097/00129039-200412000-00014
- Gougeon A. Dynamics of Follicle Development in the Human Ovary. In: Chang RJ, ed. Polycystic Ovary Syndrome. Springer New York; 1996:21-36.
- Bezerra FTG, Dau AMP, Van Den Hurk R, Silva JRV. Molecular characteristics of oocytes and somatic cells of follicles at different sizes that influence in vitro oocyte maturation and embryo production. Domest Anim Endocrinol. Jan 2021;74:106485. doi:10.1016/j.domaniend.2020.106485
- Son WY, Chung JT, Dahan M, Reinblatt S, Tan SL, Holzer H. Comparison of fertilization and embryonic development in sibling in vivo matured oocytes retrieved from different sizes follicles from in vitro maturation cycles. J Assist Reprod Genet. Jun 2011;28(6):539-44. doi:10.1007/s10815-010-9527-z
- Trounson A, Anderiesz C, Jones G. Maturation of human oocytes in vitro and their developmental competence. Reproduction. Jan 2001;121(1):51-75. doi:10.1530/rep.0.1210051
- Oldfield AL, Vanden Brink H, Carter FE, Jarrett BY, Lujan ME. Obesity is associated with alterations in antral follicle dynamics in eumenorrheic women. Hum Reprod. Mar 1 2023;38(3):459-470. doi:10.1093/humrep/dead007
- Baerwald AR, Adams GP, Pierson RA. Characterization of ovarian follicular wave dynamics in women. Biology of reproduction. Sep 2003;69(3):1023-31. doi:10.1095/biolreprod.103.017772
- Baerwald AR, Adams GP, Pierson RA. A new model for ovarian follicular development during the human menstrual cycle. Fertil Steril. Jul 2003;80(1):116-22. doi:10.1016/s0015-0282(03)00544-2
- Vanden Brink H, Chizen D, Hale G, Baerwald A. Age-related changes in major ovarian follicular wave dynamics during the human menstrual cycle. Menopause (New York, NY). Dec 2013;20(12):1243-54. doi:10.1097/GME.0b013e31828cfb62
- Weghofer A, Kushnir VA, Darmon SK, et al. Age, body weight and ovarian function affect oocyte size and morphology in non-PCOS patients undergoing intracytoplasmic sperm injection (ICSI). PLoS One. 2019;14(10):e0222390. doi:10.1371/journal.pone.0222390
- Morales C, García-Pardo L, Reymundo C, Bellido C, Sánchez-Criado JE, Gaytán F. Different patterns of structural luteolysis in the human corpus luteum of menstruation. Hum Reprod. Oct 2000;15(10):2119-28. doi:10.1093/humrep/15.10.2119
- Daghash SM, Yasin NAE, Abdelnaby EA, Emam IA, Tolba A, Abouelela YS. Histological and hemodynamic characterization of corpus luteum throughout the luteal phase in pregnant and non-pregnant buffalos in relation to nitric oxide levels based on its anatomical determination. Front Vet Sci. 2022;9:896581. doi:10.3389/fvets.2022.896581
- Bottomley C, Bourne T. Diagnosis and management of ovarian cyst accidents. Best practice & research Clinical obstetrics & gynaecology. Oct 2009;23(5):711-24. doi:10.1016/j.bpobgyn.2009.02.001
- Ellenberger C, Müller K, Schoon HA, Wilsher S, Allen WR. Histological and immunohistochemical characterization of equine anovulatory haemorrhagic follicles (AHFs). Reprod Domest Anim. Jun 2009;44(3):395-405. doi:10.1111/j.1439-0531.2008.01085.x
- Bashir ST, Baerwald AR, Gastal MO, Pierson RA, Gastal EL. Follicle growth and endocrine dynamics in women with spontaneous luteinized unruptured follicles versus ovulation. Hum Reprod. Jun 1 2018;33(6):1130-1140. doi:10.1093/humrep/dey082
- Dal J, Vural B, Caliskan E, Ozkan S, Yucesoy I. Power Doppler ultrasound studies of ovarian, uterine, and endometrial blood flow in regularly menstruating women with respect to luteal phase defects. Fertil Steril. Jul 2005;84(1):224-7. doi:10.1016/j.fertnstert.2004.12.059
- Sun BZ, Kangarloo T, Adams JM, et al. Healthy Post-Menarchal Adolescent Girls Demonstrate Multi-Level Reproductive Axis Immaturity. J Clin Endocrinol Metab. Feb 1 2019;104(2):613-623. doi:10.1210/jc.2018-00595
- Bashir ST, Gastal MO, Tazawa SP, et al. The mare as a model for luteinized unruptured follicle syndrome: intrafollicular endocrine milieu. Reproduction. Mar 2016;151(3):271-83. doi:10.1530/rep-15-0457
- Xu F, Hazzard TM, Evans A, Charnock-Jones S, Smith S, Stouffer RL. Intraovarian actions of anti-angiogenic agents disrupt periovulatory events during the menstrual cycle in monkeys. Contraception. Apr 2005;71(4):239-48. doi:10.1016/j.contraception.2004.12.017
- Murphy MJ, Halow NG, Royer PA, Hennebold JD. Leukemia Inhibitory Factor Is Necessary for Ovulation in Female Rhesus Macaques. Endocrinology. Nov 2016;157(11):4378-4387. doi:10.1210/en.2016-1283
- Walker CA, Bjarkadottir BD, Fatum M, Lane S, Williams SA. Variation in follicle health and development in cultured cryopreserved ovarian cortical tissue: a study of ovarian tissue from patients undergoing fertility preservation. Human fertility (Cambridge, England). Jul 2021;24(3):188-198. doi:10.1080/14647273.2019.1616118
- Ting AY, Yeoman RR, Lawson MS, Zelinski MB. Synthetic polymers improve vitrification outcomes of macaque ovarian tissue as assessed by histological integrity and the in vitro development of secondary follicles. Cryobiology. Aug 2012;65(1):1-11. doi:10.1016/j.cryobiol.2012.04.005
 BACK TO TOP
BACK TO TOP