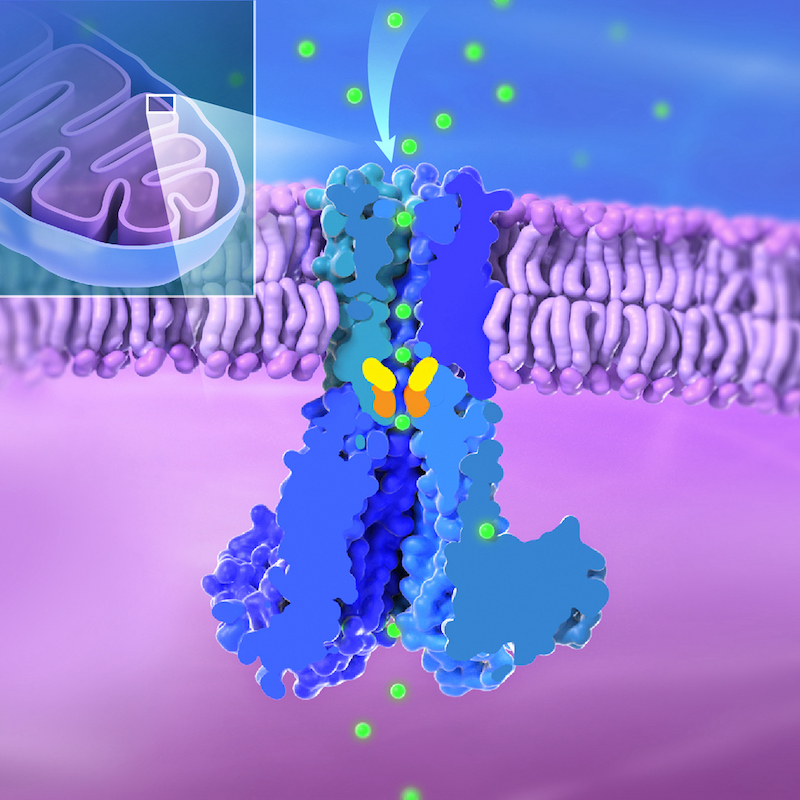
Credit: Ethan Tyler, NIH Medical Arts; Louis T.F. Lai, Ph.D., Matthies Lab, NICHD
Researchers at the National Institutes of Health have developed a model of how mitochondria—the energy producers in cells—obtain the essential mineral magnesium through the Mitochondrial RNA Splicing 2 (MRS2) channel. Their findings, published in Nature Communications, provide foundational knowledge on a process needed for healthy muscles and bones, nerve function, and immunity.
Background
The mineral magnesium is an essential nutrient that is obtained through the diet, including foods such as leafy greens, legumes, bananas, nuts, seeds, milk, and some fatty fish. Magnesium is required for many biological processes, including the production of cellular energy called adenosine triphosphate (ATP), DNA replication, and protein synthesis and stability. Magnesium also serves as a co-factor for hundreds of enzymes. Dysregulation of magnesium can result in a variety of diseases, including muscle and bone diseases, cardiac conditions, immunodeficiency, Parkinson’s disease, and cancer. Magnesium deficiency is common and associated with a higher risk of cardiovascular and gastrointestinal issues.
In mitochondria—the organelle in cells where energy is produced and metabolic processes occur—magnesium passes through the MRS2 channel. However, researchers are unsure precisely how magnesium transport is regulated through this channel.
Results
In a new study from NICHD’s Unit on Structural Biology, researchers used cryo-electron microscopy to unveil the structure of human MRS2 in the presence and absence of magnesium. The team found that MRS2 is a five-subunit structure, i.e., pentameric channel, with one central pore that magnesium passes through. They identified two key areas, called R332 and M336, that serve as gates to regulate entry of magnesium.
In addition to the known “ion selectivity filter” at the entrance of the pore, which pulls in positively charged magnesium ions, the authors found a variety of negatively charged residues, which can direct the magnesium ions. The team also identified new interactions between the channel’s subunits. All these features are involved in regulating the channel’s function.
Significance
The findings are among the first published studies to identify the structure of human MRS2 and to explain how magnesium transport is regulated. Such knowledge can benefit researchers who study conditions or diseases where magnesium is dysregulated in the body.
Reference
Lai L.T.F., Balaraman J., Zhou F., and Matthies D. Cryo-EM structures of human magnesium channel MRS2 reveal gating and regulatory mechanisms . Nature Communications DOI: 10.1038/s41467-023-42599-3 (2023)

 BACK TO TOP
BACK TO TOP