Tool has the potential to assist with preterm birth, labor management and clinical decision-making
Researchers funded by the National Institutes of Health have developed a new imaging tool, called electromyometrial imaging (EMMI), to create real-time, three-dimensional images and maps of contractions during labor. The non-invasive imaging technique generates new types of images and metrics that can help quantify contraction patterns, providing foundational knowledge to improve labor management, particularly for preterm birth. The small study is supported in part by NIH’s Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD) through its Human Placenta Project and other programs. The findings are published in Nature Communications.
“EMMI has the potential to answer critical questions about uterine contractions and will help us better understand what occurs during pregnancy and labor,” said Diana W. Bianchi, M.D., NICHD Director. “With additional research, the tool may potentially predict who is at risk to deliver prematurely or whose labor pattern will eventually result in the need for a cesarean section delivery. This will also help care providers evaluate whether a treatment or intervention is working.”
The study team, led by Yong Wang, Ph.D., and Alan Schwartz, M.D., Ph.D., at Washington University in St. Louis, and Alison Cahill, M.D., at the University of Texas at Austin, initially developed EMMI using a sheep model and reported their findings in Science Translational Medicine. In the new study, the team tailored EMMI for human clinical use and tested it among a group of 10 women with healthy pregnancies. Current clinical methods to measure contractions (i.e., tocodynamometry and an intrauterine pressure catheter) can only provide limited details, such as contraction duration and intensity, while also being invasive.
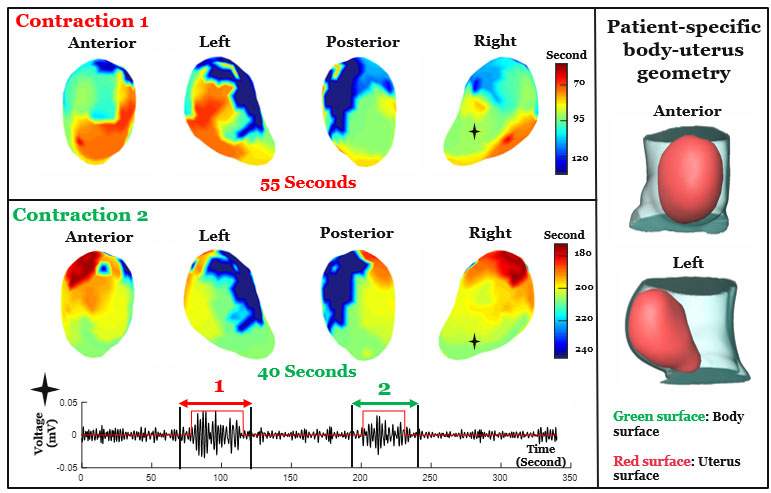
EMMI integrated two types of non-invasive scans—a fast anatomical MRI to obtain an image of the uterus (which can be taken during early term pregnancy, or 37 weeks gestation), and a multi-channel surface scanning electromyogram that uses sensors placed along the belly to measure contractions during labor. These data are then combined and processed into three-dimensional uterine maps, with warm colors denoting areas of the uterus that are activated earlier in a contraction, cool colors indicating areas that are activated later and gray areas showing inactive regions. A sequence of maps is generated over time, creating a visual timelapse that shows where contractions start, how they spread and/or synchronize, and potential patterns that are associated with a typical pregnancy versus one with complications.
Credit: Washington University in St. Louis
EMMI maps were also used to develop metrics to describe uterine contractions. The maximal activation ratio, for example, measures the total surface area of the uterus that becomes electrically active during an individual contraction. The activation curve slope measures the rate of uterine electrical activation. The fundal early activation ratio helps quantify the region that generates contractions to dilate the cervix.
Results from the pilot study also bring clarity on a longstanding question on how contractions begin—EMMI data suggest there is no fixed, pacemaker-like region in the uterus that initiates labor. The study team observed varied patterns of contractions and metrics among the 10 study participants, with some similarities between women who had never given birth and those who had. However, more research is needed to confirm and expand upon these observations.
EMMI offers new possibilities for better understanding human labor and facilitating the development of optimized, patient-specific interventions. The authors note that an EMMI contraction atlas generated from healthy pregnancies can serve as a resource to understand and diagnose preterm labor and possibly identify patients who would benefit from an induction versus those who may need a cesarean section.
REFERENCE:
Wang, H., Wen, Z., et. al. Noninvasive electromyometrial imaging of human uterine maturation during term labor. Nature Communications 10.1038/s41467-023-36440-0 (2023)

 BACK TO TOP
BACK TO TOP